Chapter 6 : Thermodynamics.
chemistry
Answer the following Questions.
1. Choose the correct answer. A thermodynamic state function is a quantity
(i) used to determine heat changes
(ii) whose value is independent of path
(iii) used to determine pressure volume work
(iv) whose value depends on temperature only
Solution :
A thermodynamic state function is a quantity Whose value is independent of a path. Functions like p, V, T etc. depend only on the state of a system and not on the path.
Hence, alternative (ii) is correct.
2. For the process to occur under adiabatic conditions, the correct condition is:
(i) ∆T = 0
(ii) ∆p = 0
(iii) q = 0
(iv) w = 0
Solution : A system is said to be under adiabatic conditions if there is no exchange Of heat between the system and its surroundings. Hence, under adiabatic conditions, q = 0.
Therefore, alternative (iii) is correct,
3. The enthalpies of all elements in their standard states are:
(i) unity
(ii) zero
(iii) < 0
(iv) different for each element
Solution : The enthalpy of all elements in their standard state is zero. Therefore, alternative (ii) is correct
4. ΔU⊖ of combustion of methane is −X kJ mol−1. The value of ΔH⊖ is
(i)=ΔU⊖
(ii) >ΔU⊖
(iii) <=ΔU⊖
(iv) = 0
Solution :
SinceΔHθ=ΔUθ+ΔngRT and ΔUθ=−Xkmol−1
ΔHθ=(−X)+ΔngRT
⇒△Hθ<ΔUθ
Therefore, alternative (iii) is correct.
5. The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are, –890.3kJmol−1−393.5kJmol−1, and −285.8kJmol−1
respectively. Enthalpy of formation of CH4 will be
(i)−74.8kJmol−1
(ii)−52.27kJmol−1
(iii)+74.8kJmol−1
(iv)+52.26kJmol−1
Solution :
According to the question,
(i)CH4(g)+2O2(g)⟶CO2(z)+2H2O(g)ΔH=−890.3kJmol−1
(ii)C(x)+O2(y)⟶CO2(g)ΔH=−393.5kJmol−1
(iii)2H2(g)+O2(z)⟶2H2O(g)
ΔH=−285.8kJmol−1
Thus, the desired equation is the one that represents the formation of CH4(g) i.e..,
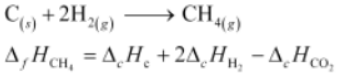
= [-395.5 + 2(-285.8) – (-890.3)] kJ Mol-1
= -74.8 kJ Mol-1
∴ Enthalpy of formation of CH4(g)=−74.8kJmol−1 Hence, alternative (i) is correct.
6. A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be
(i) possible at high temperature
(ii) possible only at low temperature
(iii) not possible at any temperature
(iv) possible at any temperature
Solution :
For a reaction to be spontaneous, ΔG should be negative.
ΔG = ΔH – TΔS
According to the question, for the given reaction,
ΔS = positive
ΔH = negative (since heat is evolved)
⇒ ΔG = negative
Therefore, the reaction is spontaneous at any temperature.
Hence, alternative (iv) is correct.
7. In a process, 701 J of heat is absorbed by a system and 394 J of work is done by the system. What is the change in internal energy for the process?
Solution :
According to the first law of thermodynamics,
ΔU = q + W (i)
Where,
ΔU = change in internal energy for a process
q = heat
W = work
Given,
q = + 701 J (Since heat is absorbed)
W = –394 J (Since work is done by the system)
Substituting the values in expression (i), we get
ΔU = 701 J + (–394 J)
ΔU = 307 J
Hence, the change in internal energy for the given process is 307 J.
8. The reaction of cyanamide,NH2CN(s) with dioxygen was carried out in a bomb calorimeter, and ∆U was found to be –742.7kJmol−1 at 298 K. Calculate enthalpy change for the reaction at 298 K.
NH2CN(g) + 3/2O2(g)→N2(g)+CO2(g)+H2O(l)
Solution :
Enthalpy change for a reaction (ΔH) is given by the expression,
ΔH = ΔU + ΔngRT
Where,
ΔU = change in internal energy
Δng = change in number of moles
For the given reaction,
Δng = ∑ng (products) – ∑ng (reactants)
= (2 – 1.5) moles
Δng = 0.5 moles
And,
ΔU = –742.7 kJ mol–1
T = 298 K
R = 8.314 × 10–3 kJ mol–1 K–1
Substituting the values in the expression of ΔH:
ΔH = (–742.7 kJ mol–1) + (0.5 mol) (298 K) (8.314 × 10–3 kJ mol–1 K–1)
= –742.7 + 1.2
ΔH = –741.5 kJ mol–1
9. Calculate the number of kJ of heat necessary to raise the temperature of 60.0 g of aluminium from 35°C to 55°C. Molar heat capacity of Al is 24 Jmol−1K−1.
Solution :
From the expression of heat (q) q=m⋅ c. ΔT
Where,
c= molar heat capacity
m= mass of substance
ΔT= change in temperature
Substituting the values in the expression of q:
q=(60/27mol)(24Jmol−1K−1)(20K)
q=1066.7J
q=1.07k
10. Calculate the enthalpy change on freezing of 1.0 mol of water at10.0°C to ice at
–10.0°C.ΔfusH=6.03kJmol−1 at 0∘C
Cp[H2O(I)]=75.3Jmol−1K−1
Cρ[H2O(s)]=36.8Jmol−1K−1
Solution :
Total enthalpy change involved in the transformation is the of the following changes:
(a) Energy change involved in the transformation of 1 mol of water at 10∘ C to 1 mol of water at 0 C.
(b) Energy change involved in the transformation of 1 mol of water at 0∘ to 1 mol of ice at 0∘C
(c) Energy change involved in the transformation of 1 mol of ice at0∘C to 1 mol of ice at−10∘C.
ΔH= Cp[ H2OCl]ΔT+ ΔH fivering + Cρ[H2O(s)]ΔT
=(75.3] mol−1K−1)(0−10)K + (−6.03 × 103Jmol−1)+(36.8] mol−1 K−1 )(−10 −0)K
=−7533 mol−1 − 6030Jmol −1− 368Jmol−1
=−7151J mol−1
=−7.151kJmol−1
Hence, the enthalpy change involved in the transformation is−7.151kJmol−1.
11. Enthalpy of combustion of carbon to CO2 is –393.5−7.151kJmol−1 . Calculate the heat released upon formation of 35.2 g ofCO2 from carbon and dioxygen gas.
Solution :
Formation of CO2 from carbon and dioxygen gas can be represented as:
C(s) + O2(g)⟶CO2(g)
ΔfH=−393.5kJmol−1
(1 mole =44g) Heat released on formation of 44gCO2=−393.5kJmol−1
∴ Heat released on formation of 35.2gCO2
=−314.8kJmol−1
12. Enthalpies of formation of CO(g), CO2(g), N2O(g) and N2O4(g) are −110,−393,81 and 9.7kJmol−1 respectively.
Find the value of ∆H for the reaction:
N2O4(g)+3CO(g)→N2O(g+3CO2(g]
Solution :
ΔrH for a reaction is defined as the difference between ΔH value of products and ΔH value of reactants.
Δ,H=∑Δ,H( products )−∑ΔfH( reactants )
For the given reaction,
N2O4(g) + 3CO(g)⟶ N2O(g) + 3CO2(g)
ΔrH=[ {ΔfH(N2O)+3ΔJH(CO2)}−{ΔfH (N2O4) + 3ΔjH(CO)} ]
Substituting the values ofΔH for N2O,CO2,N2O4 , and CO
From the question, we get:
ΔrH=[ { 81kJmol−1 + 3(−393)kJmol−1} − {9.7kJmol −1+3(−110) kJmol−1}]
ΔrH=−7777kJmol−1
Hence, the value ofΔrH
for the reaction is −777.7 kJmol−1.
13. Given
N2(g) + 3H2(g) ⟶ 2NH3(y);
ΔrHθ=−92.4kJmol−1
What is the standard enthalpy of formation ofNH3 gas?
Solution : Standard of formation of a compound is the charge in enthalpy that takes place during the formation of 1 mole Of a substance in its standard form from its constituent elements in their standard state.
Re-writing the given equation for 1 mole of NH3(g).
1/2N2(g)+3/2H2(g)⟶NH3(g)
∴ Standard enthalpy of formation of NH3(g)
=1/2 ΔrHθ = 1/2(−92.4 kJmol−1 )= −46.2kJmol−1
14. Calculate the standard enthalpy of formation ofCH3OH(l) from the following data:
CH3OH(l)+3/2O2(g]→CO2(g)+2H2O(l): Δ,H∘=−726kJmol−1
C(graphite) +O2(g)→CO2(g]: ΔeH=−393kJmol−1
H2(g)+1/2O2(g)→H2O(1); Δ,H=−286kJmol−1
Solution :
The reaction that takes place during the formation ofCH3OH(l) can be written as:
C(s) + 2H2O(g) + 1/2O2(G), ⟶ CH3OH(η)(1)
The reaction (I) can be obtained from the given reactions by following the algebraic calculations as:
Equation (ii) + 2 × equation (iii) – equation (i)
ΔfHθ [CH3OH(l)] = ΔcHθ + 2ΔfHθ [H2O(l)] – ΔrHθ
= (–393 kJ mol–1) + 2(–286 kJ mol–1) – (–726 kJ mol–1)
= (–393 – 572 + 726) kJ mol–1
ΔfHθ [CH3 OH(l)] = –239 kJ mol–1
15. Calculate the enthalpy change for the process CCl4(g)→C(g)+4Cl(g) and calculated bond enthalpy of C−Cl in CCl4(g)
ΔvapHθ (CC|4) = 30.5kJmol−1 ΔfHθ(CCl4) =−135.5kJmol−1
ΔaHθ(C) = 715.0kJmol−1, where ΔaHθ is enthalpy of atomisation
Δ2Hθ(Cl2) = 242kJmol−1
Solution :
The chemical equations implying to the given values of enthalpies” are:
(1) CCl4(l) à CCl4(g) ; ΔvapHΘ = 30.5 kJmol−1
(2) C(s) à C(g) ΔaHΘ = 715 kJmol−1
(3) Cl2(g) à 2Cl(g) ; ΔaHΘ = 242 kJmol−1
(4) C(g) + 4Cl(g) à CCl4(g); ΔfHΘ
= -135.5 kJmol−1 ΔH for the process CCl4(g) à C(g) + 4Cl(g) can be measured as:
ΔH=ΔaHΘ(C) + 2ΔaHΘ(Cl2) – ΔvapHΘ–ΔfH
= (715kJmol−1) + 2(kJmol−1) – (30.5kJmol−1) – (-135.5kJmol−1)
Therefore, H= 1304kJmol−1
The value of bond enthalpy for C-Cl in CCl4(g)
= 1304/4kJmol−1
= 326 kJmol−1
16. For an isolated system, ∆U = 0, what will be ∆S ?
Solution :
ΔS will be positive i.e., greater than zero
Since ΔU = 0, ΔS will be positive and the reaction will be spontaneous.
17. For the reaction at 298 K,
2A + B → C
∆H = 400kJmol−1
and ∆S = 0.2kJmol−1
At what temperature will the reaction become spontaneous considering ∆H and ∆S to be constant over the temperature range.
Solution :
From the expression
ΔG= ΔH−TΔS
Assuming the reaction at equilibrium,δ
T for the reaction would be:
T=(ΔH−ΔG)1/ΔS=ΔH/ΔS(ΔG=0 at equilibrium)
=400kJmol−1 0.2 kJK−1 mol−1 T=2000K
For the reaction to be spontaneous,ΔG must be negative. Hence, for the given reaction to be spontaneous, T should be greater than 2000 K.
18. For the reaction,2Cl(g)→Cl2(g) , what are the signs of ∆H and ∆S ?
Solution :
∆H and ∆S are negative
The given reaction represents the formation of chlorine molecule from chlorine atoms. Here, bond formation is taking place. Therefore, energy Is being released. Hence ∆H is negative.
Also, two moles of atoms have more randomness than one mole of a molecule. Since spontaneity is decreased, ∆S is negative for the given reaction.
19. For the reaction
2A(g)+B(g)→2D(g)ΔUe=−10.5kJ and ΔS∘=−44.1JK−1
Calculate ΔG⊖ for the reaction, and predict whether the reaction may occur spontaneously
Solution :
For the given reaction,
2A(g)+B(g)→2D(g)Δηg=2−(3)=−1 mole
Substituting the value of ΔUθ
in the expression of ΔH:
ΔHθ=ΔUθ+ΔngRT
=(−10.5kJ)−(−1)(8.314×10−3 kJK−1 mol−1) (298K) = −10.5kJ−2.48kJΔH⊖=−12.98kJ
Substituting the values ofΔH⊖ and ΔS⊖ in the expression of ΔG⊖
ΔGθ= △Hθ−TΔSθ
=−12.98kJ − ( 298K) (−44.1JK−1) = −12.98kJ + 13.14 kJ ΔG⊖ =+0.16kJ
SinceΔGθ for the reaction is positive, the reaction will not occur spontaneously.
20. The equilibrium constant for a reaction is 10. What will be the value of ∆G⊖ ? R = 8.314JK−1 mol−1
T = 300 K.
Solution :
From the expression, ΔGθ= −2.303 RT logk eq
ΔGθ for the reaction,
=(2.303) (8.314JK−1mol−1) (300K) log10=−5744.14Jmol−1
=−5.744kkmol−1
21. Comment on the thermodynamic stability of NO(g) , given
12N2(g)+12O2(g)→NO(g);
ΔrH⊖= 90kJmol−1NO(g) + 12O2(g)→NO2(g):
ΔrHe= −74kJmol−1
Solution :
The positive value of ΔrH indicates that heat is absorbed during the formation of NO(g), j. This means that NO(g) has higher than the reactants(N2 and O2) .
Hence, NO(g) is unstable. The negative value ofΔrH
H indicates that heat is evolved during the formation ofNO2(g) from NO(g) and O2(g)
. The product,NO2(g) is stabilized with minimum energy.
Hence, unstableNO(g) changes to unstableNO2(g).
22. Calculate the entropy change in surroundings when 1.00 mol ofH2O(l) is formed under standard conditions.ΔHθ=−286kJ mol−1
Solution :
It is given that 286 kJmol−1 of heat is evolved the formation of 1 mol ofH2O(l).
Thus, an equal amount of heat will be absorbed by the surroundings.
qsurr= +286 kJmol−1
Entropy change(ΔSsurr) for the surroundings = qsurr / 7
=286kJmol−1/ 298k
∴ΔSsurt =959.73 Jmol−1K−1

